
Read the next episode: “Building a Faster, Stronger Human“
Read the previous episode: “The Future of Birth Control Means Facing Up to Its Sexist Past“

Debojyoti Chakraborty smiles at me from his laboratory in New Delhi. Through the grainy resolution of our video call, I can just make out a button-up shirt and short black hair atop a boyish face; in the background, graduate students in lab coats drift in and out of shot. The CSIR-Institute of Genomics & Integrative Biology, where his lab is based, experiments with reprogramming adult tissue to take on the characteristics of embryonic stem cells, which can then grow into over 200 other cell types.
Chakraborty’s own research team is focused on two human diseases: a rare type of encephalitis and sickle-cell anemia. The latter is an inherited blood disorder, widespread across sub-Saharan Africa and among certain castes and tribal groups in India, in which the hemoglobin protein in red blood cells takes on an abnormal shape. This prevents the blood from carrying oxygen efficiently, leading to organ damage and severe pain. Because of the demographics of its genetic carriers, it disproportionately impacts poorer people. With no cure, those affected by the condition require medication throughout their lives. Unless, of course, a more permanent solution can be discovered.
Chakraborty’s laboratory is one of the best equipped in India to conduct this kind of research. He has a team of four postgraduate students; a technician and a handful of project assistants; a fully-equipped cell culture and molecular biochemistry lab; a zebrafish facility; and access to the vast datasets of genetic sequences available through the institute’s mainframe.
Ultimately their hope is to develop a “one-size-fits-all” cure for sickle-cell: one that can be produced in bulk with government support, drastically reducing the overall cost. “The good thing is that it’s a single mutation which causes this disorder,” Chakraborty says. “And if it’s a single mutation it’s much easier to target using CRISPR, so that’s what we’re trying to do.”
CRISPR, more fully known as CRISPR-Cas9, is a gene-editing technology that allows for targeted genetic editing with an accuracy never seen before. Since its discovery in 2013, researchers around the world have started working to cure many human diseases that are currently either difficult to treat, or are entirely untreatable.
Still, “easier,” as Chakraborty describes it, is subjective. The costs involved in CRISPR-Cas9 treatments are huge–and it’s hard not to question whether the huge promise of this revolutionary new technology will be available to those who need it most.

As a species, we have achieved mastery over the external environment more quickly than we have over our inner workings. The germ theory of disease was only conclusively demonstrated by Louis Pasteur in the early 1860s; before then most explanations were built on crude metaphors at best (“bad air,” “poisonous insects”) and sheer quackery at worst.
In the short time that effective medical technology has existed, it has augmented our ability to deal with the many diseases, disorders, and miscellaneous ailments that can compromise the human body. Just as technology that thinks for us enhances our ability to process information beyond our natural capacity, health inventions like the contraceptive pill allow us to control, slow, or even push back sickness and death.
In rich economies, where basic threats to health like unclean water or bacterial infection are under relative control, there are now two main frontiers for medical research. The first is chronic conditions like cancer, heart disease, and diabetes; the other is inherited genetic disorders. This is why CRISPR has so many scientists so excited.
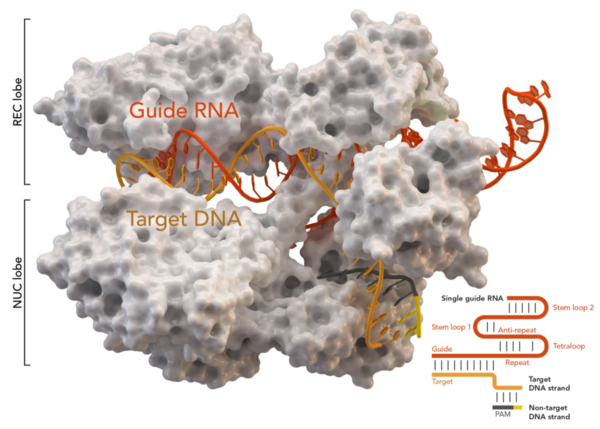
CRISPR stands for “Clustered Regularly Interspaced Short Palindromic Repeats,” short sequences of genetic code in a bacterium’s genome interspaced by samples of viruses which have attacked the bacteria in the past. Like a poster showing the face of a wanted criminal, the bacterial defence systems carry CRISPR sections around until they find a match, latching onto the enemy virus and deploying an enzyme, Cas9, that cuts it into pieces. Crucially, scientists can then substitute new sections of DNA in those gaps, making CRISPR-Cas9 the perfect tool for identifying, removing, and replacing individual genes with remarkable precision in any living thing.
The potential uses are almost limitless. A few currently active projects include sterilizing mosquitoes that transmit malaria, breeding abnormally muscular dogs for military and hunting use, and creating peanuts that won’t cause dangerous allergic reactions. It offers a way to fix currently incurable conditions like muscular dystrophy, cystic fibrosis, and Huntington’s chorea, as well as deadly transmitted diseases including HIV/AIDS.
But this is likely only for those who are able to afford it. Since these gene therapy treatments must be tailored to the DNA of each individual patient, overhead costs run into the hundreds of thousands–or even millions–of dollars per treatment. This is likely to be the case for at least the next decade, according to Jim Kozubek, a science writer and author of Modern Prometheus: Editing the Human Genome with Crispr-Cas9.
Kozubek explained some of the reasons for the high price tag: “If you have a living human, how are you going to get CRISPR into all of their cells, or some of their cells in particular?” he told me. “One way to do it is ex vivo: you take T-cells or blood cells out of the patient’s body, treat their cells, and then return the cells back to them–that’s called autologous cell therapy. Or you can use a virus to do in vivo therapy, which is to scale up CRISPR into a retrovirus or adeno-associated virus and then dose a human with thousands or millions of copies of the virus to carry CRISPR into their living cells and treat them that way.”
Either method will cost somewhere in the tens of thousands of dollars per person. On top of that, there’s the cost of hospitalization to administer the treatment, followed by careful monitoring and a host of other steps to supervise recovery and manage risk. The final bill will be somewhere on a par with the average cost of a new house in the U.S.
“Right now it can’t be very exciting for anyone that doesn’t have high-end healthcare,” Kozubek observes. There’s a clear disconnect between how easy CRISPR makes it to edit cells and the prohibitive cost of using it–and, in turn, the ability of normal people to access it.

On August 30, 2017, the U.S. Food and Drug Administration approved the first gene-altering leukemia treatment for humans. Marketed under the name Kymriah and produced by the pharmaceutical giant Novartis, it uses a technique developed by researchers at the University of Pennsylvania called CAR-T cell therapy. Patients’ own immune cells are engineered to attack cancerous cells in a way that would not otherwise be biologically possible, turning them into what has been called “a living drug.”
Only one treatment session with Kymriah is necessary to reverse an aggressive form of childhood leukemia that is all but untreatable other means. The price tag? $475,000. Though Kymriah is not directly related to CRISPR, its release is a good indication of how CRISPR will be brought to market–and its ultimate cost.
“If you have a piece of medical technology, in order to sell it you need to get regulatory approval,” says Jakob S. Sherkow, an associate professor of law at New York Law School and specialist in patent law in the biological sciences. “These trials are inordinately expensive at large scale. What that means is that sometimes companies are wary of spending hundreds of millions of dollars to bring their product to market if they’re not going to have some kind of exclusivity period afterwards.”
Nothing about this is unique to CRISPR, Sherkow is keen to point out. The pipeline running from discovery to clinical trials to patented product is standard procedure in the pharmaceutical industry, for better or worse. But the factors dictating how much research funding is allocated to the study of different diseases mean that conditions which affect the poor can be chronically understudied.
In 1990, the Global Forum for Health Research, which promotes health research on diseases affecting poorer nations, popularized the concept of the “10/90 gap,” where only 10 percent of global health research funding targets the countries in which 90 percent of preventable deaths occur. A 2014 University of Chicago study corroborated these findings–the global disease burden of a given condition showed no correlation whatsoever with the funding it receives. Instead, research is correlated with the “market for treatment,” the disease burden multiplied by the wealth of the nations most affected.
Right now CRISPR editing in humans is discussed in terms of treatments for either chronic or genetic diseases (eradicating malaria transmission is a notable exception). On a technical level, new treatments for diabetes could provide just as much hope in Nairobi as New York, but in practical terms, pharmaceutical companies are in the business of developing products for those who can most afford to pay for them.
Given the high price point, most CRISPR-based therapies are likely to be financed by some form of health insurance. This means that in nations that rely largely on private healthcare, insurance companies will end up being one of the main gatekeepers of public access to the technology, so their willingness to include genetic treatments in policy coverage is key.
Already there are signs that some insurers intend to do exactly the opposite. As Kozubek has noted, a handful of American insurance companies have already issued policies that specifically exclude gene therapies in order to avoid bearing the cost, a move that could set a precedent across the industry. In the U.S., sickle-cell anemia most commonly afflicts African-Americans and other communities of color, which tend to be poorer and have worse access to healthcare than less-affected communities. (Princeton anthropology professor Carolyn Rouse has argued that “sickle-cell disease funding is a form of social justice for blacks as breast cancer funding is for women.”)
But while the web of research institutions, pharmaceutical giants, and insurance companies shows what corporate control of CRISPR looks like, a network of smaller, scrappier players is coming up with a blueprint for what could be a radical alternative.

In Oakland, California, Josiah Zayner pulls a piece of sticky tape off his webcam, uncovering a lens still blurry from the adhesive. “I don’t like being spied on,” he says with a wry smile.
It’s a statement typical of a hacker, although Zayner has moved from computers into the biosciences, now identifying as part of the “biohacker” movement. After working as a research scientist for NASA’s synthetic biology program, Zayner left to found his own company, The ODIN. It sells gene engineering equipment, including a DIY-CRISPR kit for amateurs–you can modify a harmless E.coli bacteria so that it can survive on material that would normally kill it.
That might not sound spectacular, but it’s an embodiment of Zayner’s belief that for the benefits of gene editing technology to be equitably spread, ordinary people must be able to use it. In contrast to the tightly controlled clinical trials that the FDA requires, he has chosen to experiment on a subject who can forego such constraints: himself.
“If I say I want to take a patient group and run an experiment to give them bigger muscles, no institutional review board will approve that,” he explains. “But if somebody came to me and said, I need this gene in this DNA so I can inject it into myself, for me to produce it wholesale would only cost a few thousand dollars.” After a pause, he adds: “The experiments aren’t expensive, it’s just that they’re not robust, and you need someone who’s a little crazy and willing to try it.”
Of course, review boards exist for a reason, not least of which is to safeguard medical subjects from procedures that could cause them harm. The idea of deregulated, cosmetic gene editing will certainly be beyond the pale for some, and any procedure that can cause irreversible change (and possibly damage) deserves careful scrutiny.
But the DIY approach is if nothing else a counterbalance to the idea of a world of extremely efficient but mostly unaffordable genetic cures. Zayner is just one part of the growing biohacker movement across the world, many of whom are engaged in similar missions to democratize access to medicines and drive down costs.

Another of Kozubek’s concerns is the power of those who direct gene editing research–and their accountability. “There’s a movement right now of people who are super excited about the science,” he says, “but maybe are losing sight of the fact that they’re creating new inequalities in the world, or losing a sense of why they’re even doing science, in the face of the entrancing power of modifying gene code.”
Perhaps the biggest question in the ongoing debate over CRISPR is not which diseases to target. Should it be permissible to edit genetic code that will become inherited by future generations, introducing new characteristics into the gene pool forever?
When gene editing is used in a treatment for humans–like correcting abnormal hemoglobin to cure sickle-cell disease–the changes remain localized and do not affect the genetic material that the patient will pass on to their children. But as research teams in China (and more recently the U.S.) have demonstrated, it’s also possible to create mutations that will be passed on to offspring. This process, known as germline modification, is controversial for many reasons, from one-off unwanted and potentially harmful mutations to straying into the territory of eugenics.
In February 2017, the National Academy of Sciences and the National Academy of Medicine convened an international committee of scientists and ethicists to deliberate on the ethics of human gene editing. The committee’s final report concluded that, although not ready at present, editing heritable human genes could be permissible in future under certain conditions. They recommended that research focus on the treatment and prevention of disease rather than providing “enhancements” to already healthy subjects.
For some, this cautious endorsement was a step too far. Marcy Darnovsky is executive director of Genetics and Society, a nonprofit that advocates for responsible use of genetic technologies. Darnovsky has spoken widely on the social implications of gene editing, and she was critical of the report’s findings when it was released. Even as a treatment for genetic disease, she argues, the best option is to eschew germline editing in favour of existing alternatives.
“The fact is that in almost every case, if you’re a carrier of a genetic variation that would make your future child have one of these inherited diseases, you can prevent it,” she says. She suggests pre-implantation genetic diagnosis (PGD), in which embryos are created via in vitro fertilization. Ones that carry a harmful variant of a given gene are discarded, until a healthy one is found and used to initiate the pregnancy.
“It’s not ethics-free–it does raise questions about what conditions are serious enough to be eliminated from the population,” says Darnovsky. But for the appropriate cases, she believes that it’s a powerful alternative which is often left out of the discussion. “We’ve been stunned at how many media reports on the prospect of CRISPR editing for reproduction don’t even mention [PGD], though everybody in the field knows about it.”

It’s too soon to predict how readily gene editing technology will be available through smaller providers. We may or may not see “gene parlors” on Main Street, but Zayner and my other interviewees agreed that now is an important time to begin a conversation involving everyone, rather than letting big industry shape a discussion behind the scenes.
“Everyone can see that this is a technology that could, literally, reshape human beings, and reshape social relationships,” says Darnovsky. “Everyone has a stake in it, so all experts are saying that this is not just a scientific question, it’s a political and social question, so we need widespread public participation.”
Participation is also crucial to the conversation about cost of and access to healthcare–a hot topic in more than just the U.S. Nothing about this is specific to CRISPR, but as a cutting-edge treatment it illustrates the difficulty of balancing potentially huge and immediate benefits with the need for meticulous, costly trials, and a requirement for return on investment.
As a technically advanced cure for diseases that are in themselves so closely tied to modern lifestyles, gene editing technology represents a very human quest: to solve problems that we also inadvertently cause. The strands of morals, ethics, and science are tightly intertwined, and right now we’re in the process of picking them apart, choosing what to keep, what to cut out, and what to replace.
So far, there’s no miracle tool to do that work for us.

Read the next episode: “Building a Faster, Stronger Human“
Read the previous episode: “The Future of Birth Control Means Facing Up to Its Sexist Past“

How We Get To Next was a magazine that explored the future of science, technology, and culture from 2014 to 2019. The Human Machine is an eight-part series that interrogates the increasingly blurred lines between humans and machines.
